It was previously established that the stability of water polyhedra is determined by the competition of two factors. Polyhedral water clusters are convenient objects for studying the effect of proton disorder on the properties of ice-like systems. Configurations with different arrangements of hydrogen atoms (protons) in hydrogen bonds are not equivalent in their properties. In fact, this structural variety is much wider due to the proton disorder. The ability to form numerous crystalline modifications of ice and gas hydrate frameworks is a characteristic feature of water.
#Pentagonal prism free
This is the stabilizing locking of the most polarized configurations, as well as very intense amplitude oscillations of the free OH group at the end of two-bond flips. The stability of the configurations, which retained their original shape during local geometric optimization, was studied by the molecular dynamics method with a gradual increase in temperature. It is concluded that the structural stability of configurations and the features of structural transformations are basically determined by the arrangement of the homodromic hydrogen-bonded water rings. A number of statements about the topology of the hydrogen-bonded network are rigorously proved. But the main attention is paid to the concerted molecular reorientations leading to the proton shift along hydrogen bonds.
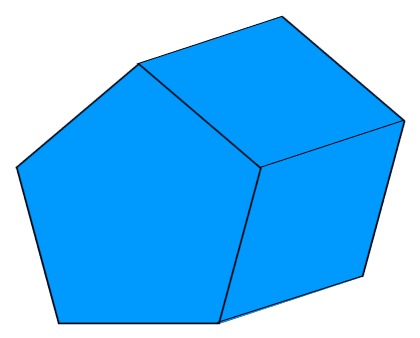
Most of the configurations changed their shapes, forming surface defects of the same type. A number of different barrier-free molecular rearrangements were observed.
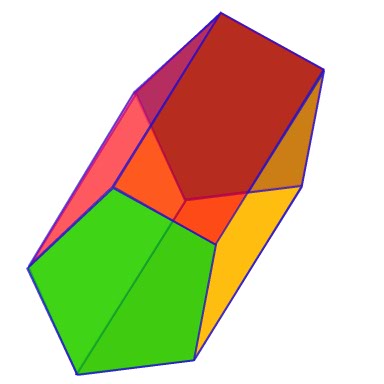
The structural stability of the weakest bonded configurations of water clusters in the form of gas hydrate cavities D and T, as well as in the form of Kelvin’s polyhedron, was tested using the local geometric optimization with the polarizable force field AMOEBA.
